Contribution #1
Rutherford after many experiments came to the conclusion that the following were true:
- An alpha particle (α=He^+2) is positive
- A Beta particle (β=e-) is negative
- A neutral particle (ϒ) is light
Contribution #2
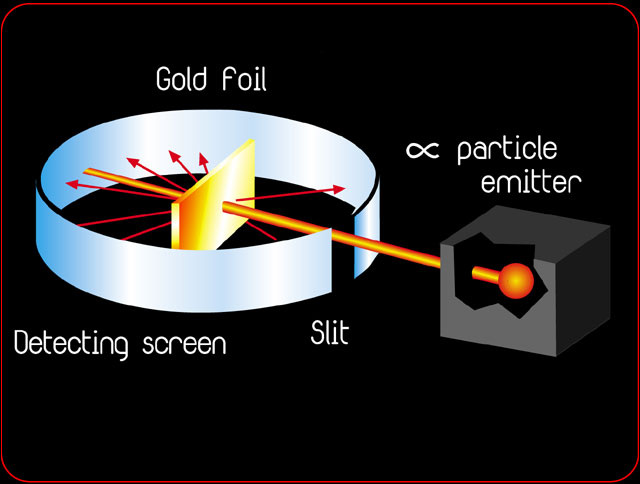
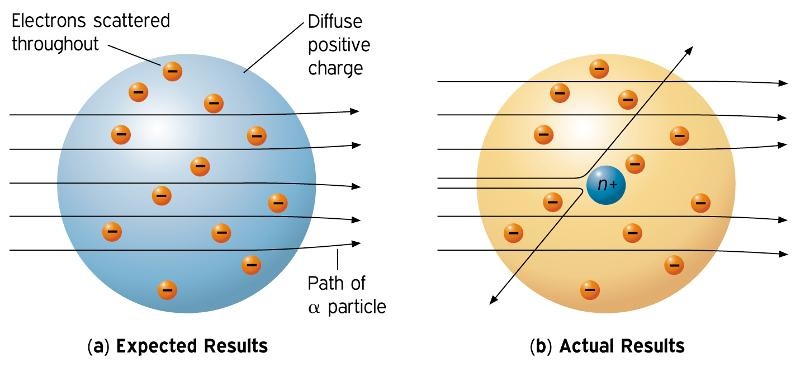
Rutherford's most famous experiment is the gold foil experiment. It consisted of shooting Alpha atoms into extremely thin gold foil and seeing where on the surrounding circular wall it hit. He believed they would all just go straight through. However, a very small amount went either off to the sides or back towards the Alpha atom stream. This he found meant that there was something in the center of the atoms, called the nucleus. This led to his nuclear model of an atom. It was the first of its kind to display the nucleus that he found while experimenting.
Significance of Contributions
Found out the charge of atoms and began the understanding of the interior of an atom.
Fun Fact
The element 'rutherfordium' was named in Rutherford’s honor.