Contribution
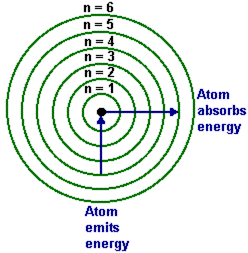
Bohr studied the structure of an atom. He suggested that electrons didn't spiral into the nucleus, but orbit in different levels. This is called the bohr model, or the quantum model. The more energy an atom gives off, the closer it is to the nucleus, the more it absorbs, the farther away. It was almost errorless when he won the Nobel Prize for it, however over the years other chemists have tweaked it a bit.
Significance of COntribution
It gave us a better understanding of why atoms bond and how they stay the same.
Fun Fact
His son Aage also won the Nobel Prize in physics.